RELEVANCE: Despite multiple clinical trials testing medications targeting dopaminergic, serotonergic, cholinergic, and gaba-ergic systems, no effective medication for methamphetamine (MA) use disorder is currently available.
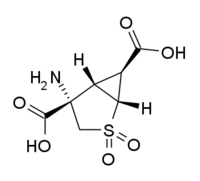
DESCRIPTION: This is an inpatient, phase I safety-interaction trial of POMA (Pomaglumetad Methionil) in participants with MA (methamphetamine) use disorder. The goal of the study is to provide the initial safety, tolerability, and pharmacokinetic (PK) data required by the FDA to advance POMA to phase 2 safety and efficacy testing. In addition, the study will investigate potential effects of POMA on a laboratory self-administration model of MA relapse. The design is a randomized, double-blind, placebo-controlled multiple ascending-dose study in 24 non-treatment seeking participants with MA use disorder.
STATUS: This study has ended and data are currently being analyzed.
